Skin Resurfacing
Skin Resurfacing
Repair signs of skin damage for a smoother, healthier-looking complexion.
Skin Resurfacing Treatments
Over 70% of consumers report feeling somewhat to extremely bothered by their imperfect skin texture and/or discoloration, according to a recent consumer survey by the American Society for Dermatologic Surgery (ASDS)¹. These conditions can often cause individuals to feel uncomfortable and self-conscious. In fact, wanting to appear more attractive and feel more confident are among the top reasons consumers are turning to cosmetic procedures.
Fractional non-ablative skin resurfacing has become a popular procedure to help treat and reduce the appearance of acne scars and other scars, striae, rosacea, dyschromia, deep wrinkles, enlarged pores, and uneven skin texture and pigmentation. This works to cosmetically improve the appearance of the skin, with low downtime and minimal discomfort for the patient when compared to surgical methods. It’s no wonder that micro-ablative skin resurfacing was the number one fastest growing non-surgical procedure in 2017. ²
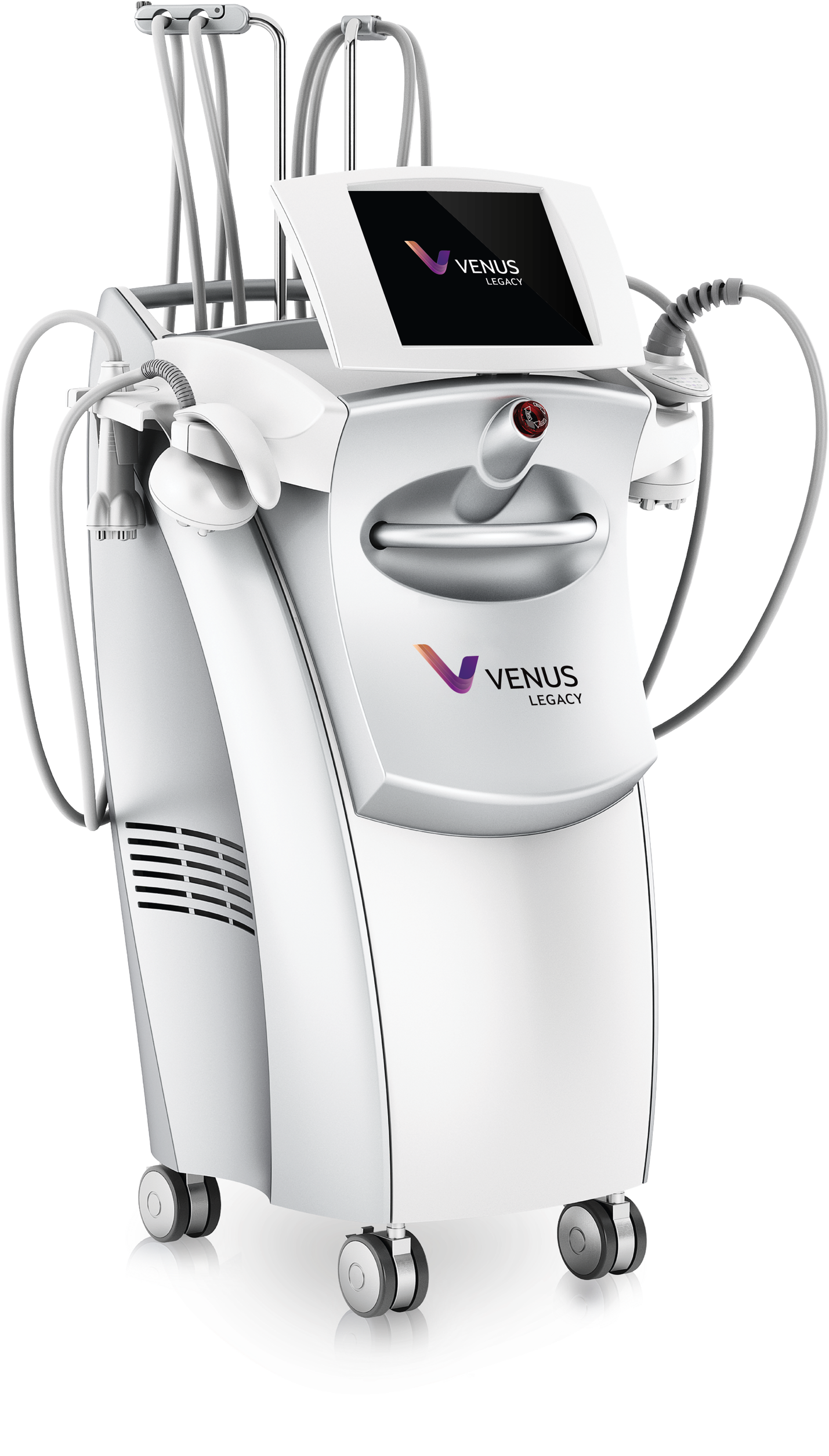
Skin Resurfacing with Venus
The non-invasive skin resurfacing systems from Venus use NanoFractional Radio Frequency (RF) technology to effectively improve the appearance of uneven skin texture and reduce other signs of skin damage without affecting the surrounding tissue. With our highly customizable treatment, finely tipped pins emit radio frequency energy to ablate the tissue in a small footprint per pin—helping to rejuvenate the appearance of the skin and resulting in a smoother, younger-looking complexion. The applicator can effectively replace all other skin resurfacing devices, offering treatments that are safe for all skin types with very low downtime.

89%

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.
89%
89%


Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.
89%

70%
Over 70% of consumers report feeling somewhat to extremely bothered by their imperfect skin texture and/or discoloration.
Safe for all skin types
NanoFractional Skin Resurfacing treatments are safe for all skin types with little downtime.
Number One Growth
Micro-ablative skin resurfacing was the number one fastest growing non-surgical procedure in 2017².
The Results Speak for Themselves
All of the before-and-after photos for our aesthetic treatments are from certified Venus providers showcasing real patient results. Images are never edited or altered.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.

Product Name™
Three word description
Name of the person
Title of person goes here

Name of the person

Title of person goes here

Name of the person
Title of person goes here

Name of the person
Title of person goes here
Title of person goes here

Name of the person

Name of the person
Title of person goes here
Three word description
Product Name™


Product Name™
Three word description
Skin resurfacing devices
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
Name of the person
Title of person goes here

Name of the person

Title of person goes here

Name of the person
Title of person goes here

Name of the person
Title of person goes here
Title of person goes here

Name of the person

Name of the person
Title of person goes here

Venus Legacy™
Three word description
FAQs
Title goes here

Three word description
Venus Versa™

Advanced versatile multi-treatment platform
Venus Versa ™ Pro
Frequently Asked Questions
BEFORE THE TREATMENT
Everything you need to know about your skin resurfacing treatment.
-
1. What devices can do skin resurfacing?
If you’re getting a skin resurfacing treatment done, your treatment provider may be using Venus Viva™ MD, Venus Versa™, and Venus Versa™ Pro.
-
2. Am I a good candidate for this treatment? / Will this treatment work for me?
The ideal candidate for radio frequency skin resurfacing is anyone who shows signs of skin damage, including deep wrinkles, scars, visible pores, stretch marks, and/or anything else that affects the skin’s texture.
For a full list of exclusion criteria, consult with your treatment provider to see if this is right for you.
-
3. I have darker skin – can I still get this treatment done?
Yes. Venus Concept’s skin resurfacing treatments use technology that’s safe for all skin tones, even darker ones.
-
4. How many sessions will I need to see results?
Most patients receive 3-4 treatments per area. The exact number will depend on each individual person and the area being treated.
-
5. How often/far apart should the treatments be?
Treatments are done 4-6 weeks apart.
-
6. Do I need to do anything to prepare for the treatment?
Make sure your skin is clean. Do not apply any lotions, creams, perfumes, powder, or makeup before your treatment session, and avoid tanning. Remove all jewelry around the area being treated. If you have excessive hair on the treatment area, shave it beforehand. It also helps to be prepared with information about your medical history—the technician will likely ask about it.
-
1. How long does a treatment session last?
Each session lasts 15-30 minutes, depending on the area being treated.
-
2. What does the treatment feel like?
You will feel tiny pins on your skin followed by a little zap. Your face will then feel warm, like a sunburn.
-
3. Will it be painful?
Venus Concept’s radio frequency skin resurfacing treatments may be slightly uncomfortable, but not painful. You’ll feel intense heat, similar to the feeling of sunburn, and your face will look red right after the treatment. For more aggressive treatments, you may get a topical numbing cream to reduce any discomfort.
-
1. How will I feel after my treatment?
Your skin will be red and feel warm afterward, like a sunburn. Follow the post care instructions given to you by your treatment provider.
-
2. Is there downtime after the treatment? / How long is recovery time?
Yes, but only for 24 hours post-treatment or up to 5 days for more aggressive treatments. Follow the post care instructions given by your provider.
-
3. How long will the results last and are they permanent?
After your final treatment, your results will continue to improve. However, one touch- up session may be needed approximately every 6 months, depending on how your skin responds.
-
4. Do I need to do anything after my treatment?
Avoid touching or scratching the treatment area. For the first 24 hours, only apply products that are expressly recommended by your treatment provider, as there are many topicals that could cause severe irritation. After 24 hours, you should apply sunscreen (at least SPF 30), preferably one without chemicals. Also avoid rigorous exercise and hot baths or massages on the treatment area for the first 2 days after your session. Make sure to keep your skin clean and protected from the sun. Avoid any sort of tanning.
DURING THE TREATMENT
AFTER THE TREATMENT
Speak To An Expert
To book a consultation or a demo, please contact us by clicking the button below and a representative will reach out to you directly.
Why Venus
Body Devices
Body Treatments
Face & Skin Devices
Face & Skin Treatments
Hair Restoration Devices
Hair Removal Devices
Legal
For more information call: (888) 907-0115 // info@venusconcept.com // 235 Yorkland Blvd., Suite 900, Toronto, ON, M2J 4Y8 Canada
For more information call: +61 416 022 096 // info.au@venusconcept.com // Suite 3.06, Level 3 20 Rodborough Road Frenchs Forest, NSW 2086, Australia