Aesthetic Body Treatment Machines
Body Treatments
With 4 in 5 Americans admitting they want to change at least one part of their body, most notably the midsection, the potential for success with aesthetic body treatment machines is significant, to say the least. Discover our full collection of medical body treatment devices to help you meet the growing demand.
Fat Reduction
was the most popular non-surgical treatment for both men and women after injectables.
More than 85%
of consumers turn to cosmetic procedures because of excess weight on their bodies.
Over $434 million
is spent on non-surgical aesthetic body procedures (skin tightening, cellulite treatments, and fat reduction).

Why You Should Be Investing in Aesthetic Body Treatment Machines
Providers must invest in a broad range of aesthetic body treatment machines to address the needs of today’s patients. From fat reduction to skin tightening and everything in between, body aesthetics procedures consistently top the list, year after year, of the most in-demand treatments among consumers. By equipping your business with our body equipment, your practice will be able to take advantage of this growing market segment.
The Results Speak for Themselves
All before-and-after photos for our body aesthetics treatments are from certified Venus providers showcasing real patient results. Images are never edited or altered.

Venus Bliss MAX™
Advanced 3-in-1 Body Shaping Solution Targeting Fat, Muscle, and Skin Body Treatments.
Venus Bliss MAX™ combines three advanced technologies to target fat, muscle, and skin in one system. The 1064 nm diode laser offers non-invasive fat reduction, FlexMAX EMS strengthens and tones muscles, and (MP)² technology reduces cellulite, leaving skin smoother and firmer-looking. All body treatments are comfortable, safe for all skin types, effective, and require no downtime.


Venus Bliss™
Advanced Non-Invasive Lipolysis Body Machines.
The Venus Bliss™ is a powerful, non-invasive lipolysis system that combines two advanced technologies in a single device. The 1064 nm diode laser targets fat reduction in the abdomen and flanks, providing patients with safe, effective body treatments with minimal discomfort. The integrated (MP)² applicator with VariPulse™ technology tightens the skin, reduces cellulite, and contours the body for a more sculpted look.


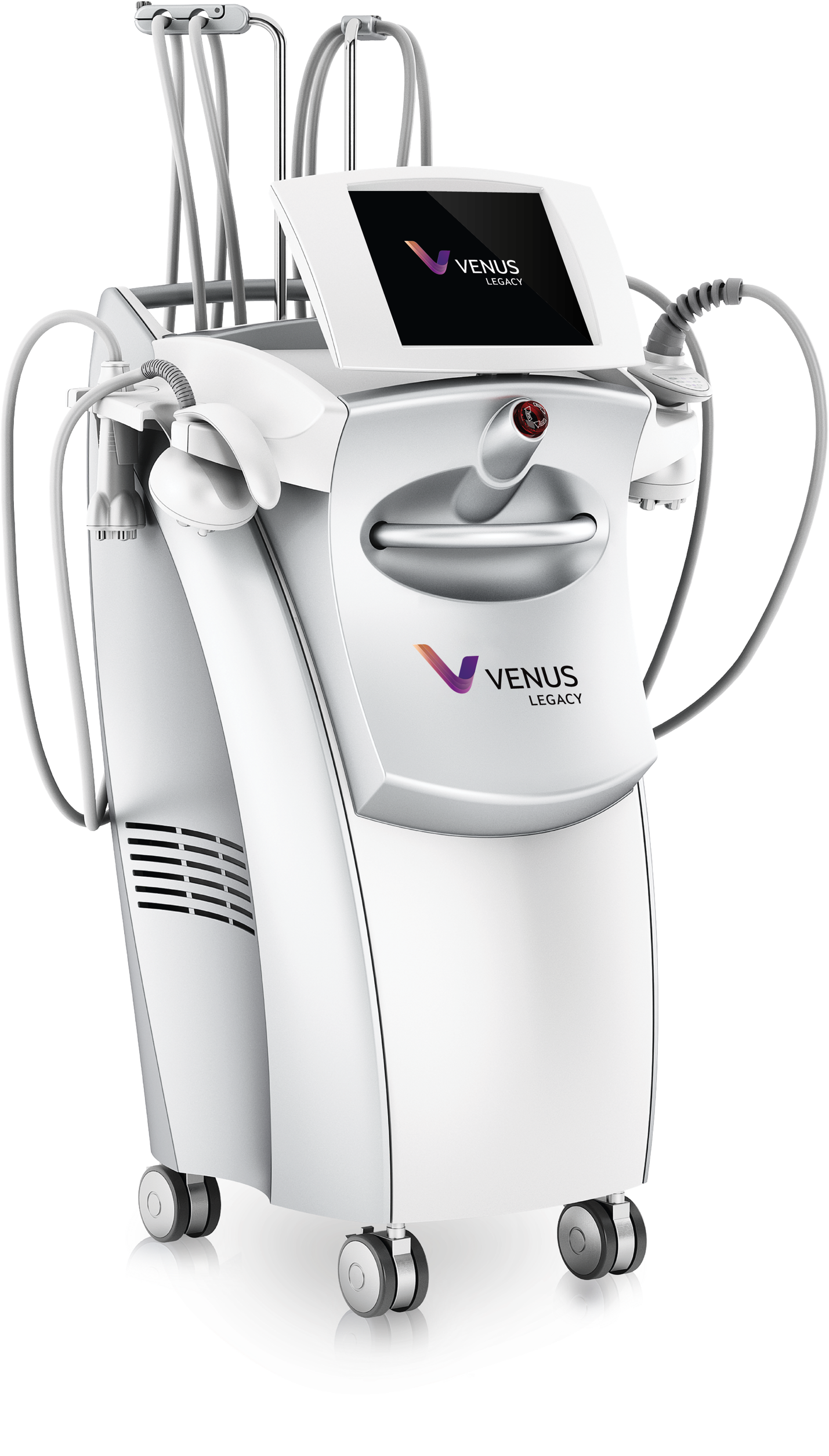
Venus Legacy™
A Full Body & Face Advanced Solution for Wrinkles, Cellulite, and Skin Tightening.
The Venus Legacy™ is a radio frequency device that treats facial and body aesthetic concerns. Powered by proprietary (MP)² technology, it combines Multi-Polar Radio Frequency with Pulsed Electromagnetic Fields to promote skin tightening and improve elasticity. Highly effective in reducing wrinkles and cellulite, treatments are comfortable, require no downtime, and are safe for all skin types.

Venus Versa™
Venus Versa™ Multi-Treatment Platform Addressing Your Top Aesthetic Needs.
The Venus Versa™ is a versatile platform designed to deliver a range of non-invasive aesthetic treatments, including photorejuvenation, acne treatment, hair removal, skin resurfacing, body contouring, cellulite reduction, skin tightening, and wrinkle reduction. It also features the exclusive TriBella™ treatment, which combines three advanced technologies to improve skin tone, firmness, and texture for a youthful appearance.


Venus Versa™ Pro
A Versatile Multi-Treatment System Designed to Meet Every Aesthetic Demand.
An upgrade to the Venus Versa™, the PRO version offers 10 applicators and 25 treatments to address an even wider range of aesthetic needs. It provides non-invasive solutions, including photorejuvenation, acne treatment, hair removal, skin resurfacing, body contouring, cellulite reduction, skin tightening, and wrinkle reduction. Boost your spa or clinic's ROI with this multifunctional device delivering top treatments on a single platform.

Venus offers a variety of aesthetic body treatment machines designed to deliver today’s most in-demand non-invasive procedures. These include versatile multi-treatment platforms that enable your clinic to provide more comprehensive body treatment plans. Depending on which body equipment you choose, treatments can include circumferential reduction, cellulite reduction, stretch mark reduction, and more.
Our Full Range of Aesthetic Body Devices
Speak To An Expert
To book a consultation or a demo, please click the button below, and a representative will contact you directly.
REFERENCES:
1. RealSelf. (2018). RealSelf Aesthetics Interest Survey. Retrieved from https://brandfolder.com/s/plg6ve-b2u2fk-11upqw
2. American Society for Aesthetic Plastic Surgery. (2018). 2018 Cosmetic (Aesthetic) Surgery National Data Bank Statistics. Retrieved from https://www.surgery.org/sites/default/files/ASAPS-Stats2018_0.pdf
3. American Society for Dermatologic Surgery. (2018). 2018 ASDS Consumer Survey on Cosmetic Dermatologic Procedures. Retrieved from https://www.asds.net/Portals/0/PDF/consumer-survey-2018-infographic.pdf
4. American Society for Aesthetic Plastic Surgery. (2017). 2017 Cosmetic Surgery National Data Bank Statistics. Retrieved from https://www.surgery.org/sites/default/files/ASAPS-Stats2017.pdf





