News & Events
Events & Tradeshows

By Josh Wiersma
•
11 Jan, 2024
Journey into the forefront of aesthetics with our highlight reel from the Venus NEXThetics event, in Texas. Attendees were the first to witness the launch of our latest advancement, Venus Versa™ Pro; offering 10 applicators, 25 treatments, and over 100+ aesthetic goals to grow your aesthetic practice. Renowned speakers, including the esteemed Cheryl Haseeb, DNP, shared invaluable insights alongside industry leaders such as Gilly Munavalli, MD, MHS, FACMS, and Scott Gerrish, MD. This is just the beginning. Stay tuned for upcoming Venus NEXThetics events, where we continue to redefine the landscape of aesthetics…

01 Feb, 2023
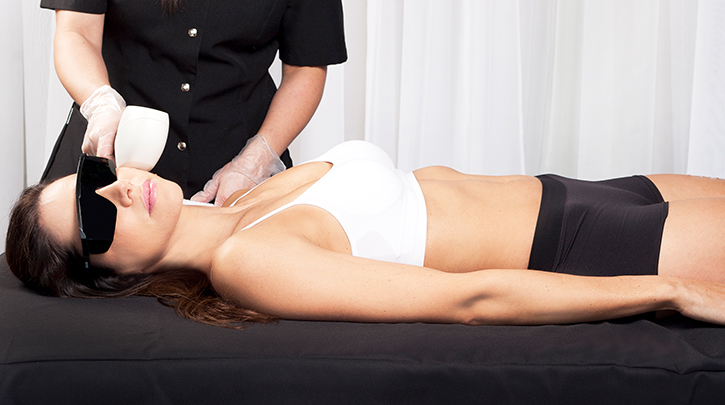
Hair removal is one of the most popular medical aesthetic procedures globally, and the trend is on the rise. To capitalize on the trend, you’ll want to offer the best in laser hair removal technology at your clinic and follow our top tips for marketing laser hair removal treatments at your practice.
Latest News
Subscribe To Our Newsletter
Catch up on news and events from the medical aesthetic industry around the globe. Join the mailing list to get the latest updates, including free webinars, practice enhancement tips, educational resources, and more.
Why Venus
Body Devices
Body Treatments
Face & Skin Devices
Face & Skin Treatments
Hair Restoration Devices
Hair Removal Devices
Legal
For more information call: (888) 907-0115 // info@venusconcept.com // 235 Yorkland Blvd., Suite 900, Toronto, ON, M2J 4Y8 Canada
For more information call: (888) 907-0115 // info@venusconcept.com // 235 Yorkland Blvd., Suite 900, Toronto, ON M2J 4T8 Canada