Wrinkle Reduction
Wrinkle Reduction
Discover our non-invasive wrinkle reduction solutions for this top, in-demand aesthetic treatment.
One of the top reasons consumers turn to cosmetic procedures is because they want to look as young as they feel, according to a recent survey by the American Society for Dermatologic Surgery (ASDS)¹. If your patients are seeing the formation of wrinkles or skin laxity related to aging, it’s common for them to want to tighten and improve their skin in order to put their best face forward. The most common sign of aging is typically the formation of wrinkles and rhytides, making it no surprise that wrinkle reduction continues to be among the most popular treatments in today's aesthetic market. The rise in non-surgical anti-aging solutions, like fillers and Botox, demonstrates how much the demand has grown. If you’re looking to provide an effective non-surgical wrinkle treatment, consider the anti-aging devices offered by Venus.
-
MORE
Venus offers a variety of different anti-aging solutions that provide non-invasive wrinkle reduction and skin tightening technology that is safe for all skin types, with zero downtime. Our multi-treatment platforms feature propriety MultiPolar Radio Frequency technology enhanced by PEMF (Pulsed Electro Magnetic Fields) for one of the most comfortable & effective treatments on the market.
Wrinkle Reduction Treatments
89%

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.
89%
89%


Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.

89%
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Lacus, sit dolor quis pharetra.
89%
Safe & No Downtime
Venus wrinkle-reduction treatments are safe and effective with zero downtime after.
0 $
Zero usage fee per treatment for the provider.
Increasing Demand
for non-surgical anti-aging solutions.
The Results Speak for Themselves
All of the before-and-after photos for our aesthetic body treatments are from certified Venus providers showcasing real patient results. Images are never edited or altered.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.

Product Name™
Three word description
Name of the person
Title of person goes here

Name of the person

Title of person goes here

Name of the person
Title of person goes here

Name of the person
Title of person goes here
Title of person goes here

Name of the person

Name of the person
Title of person goes here
Three word description
Product Name™


Product Name™
Three word description
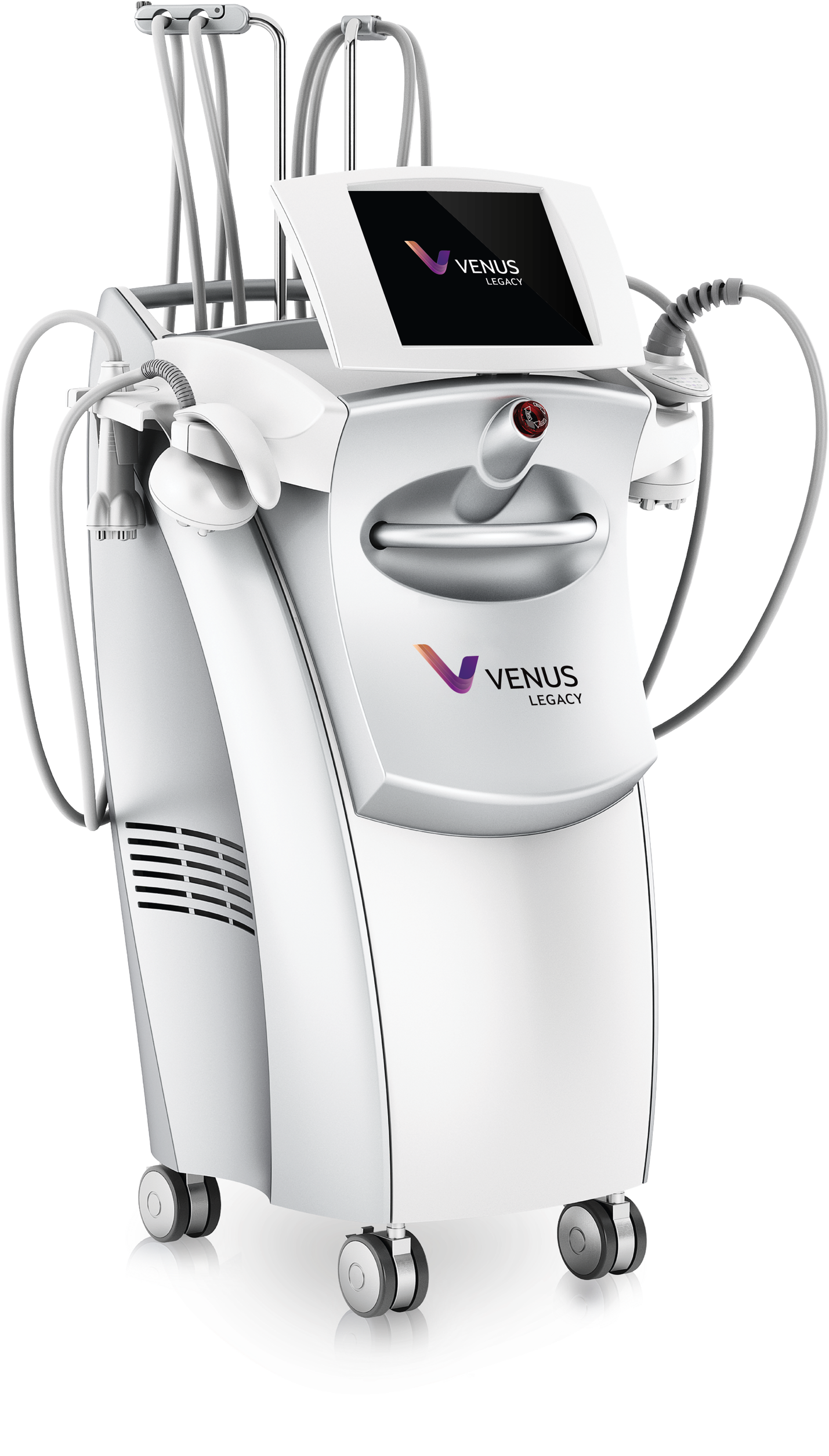
Wrinkle Reduction Devices
Venus offers a variety of different anti-aging solutions that provide non-invasive wrinkle reduction and skin tightening technology that is safe for all skin types, with zero downtime. Our multi-treatment platforms feature propriety MultiPolar Radio Frequency technology enhanced by PEMF (Pulsed Electro Magnetic Fields) for one of the most comfortable & effective treatments on the market.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
This is a paragraph. Writing in paragraphs lets visitors find what they are looking for quickly and easily.
Name of the person
Title of person goes here

Name of the person

Title of person goes here

Name of the person
Title of person goes here

Name of the person
Title of person goes here
Title of person goes here

Name of the person

Name of the person
Title of person goes here
FAQs
Title goes here
Frequently asked questions
Everything you need to know about non-invasive wrinkle treatments.
-
1. What devices can do wrinkle reduction?
If you’re getting an RF wrinkle removal treatment done, your treatment provider may be using Venus Freeze™, Venus Freeze Plus™, Venus Legacy™, Venus Versa™, or Venus Viva™ MD.
-
2. Am I a good candidate for this treatment? / Will this treatment work for me?
The best candidate is someone who is starting to lose elasticity in their skin and is showing signs of sagging, fine lines, and wrinkles.
For safety reasons, you may not be able to get this treatment if you have/are:
- Active inflammation and/or infection in the treatment area
- Current or history of cancer
- A degenerative neurologic disease or any other disease around the treatment area that’s triggered by heat (e.g. herpes)
- An Internal defibrillator/pacemaker
- Metal implants (excluding dental) in the treatment area
- Uncontrolled thyroid gland disorder
- Varicose veins
- Pregnant or are undergoing an IVF procedure
- Skin-related autoimmune diseases
- Silicone implants and/or injections in the area
-
3. I have darker skin – can I still get this treatment done?
Yes. Venus Concept’s wrinkle reduction treatments use technology that’s safe for all skin tones, even darker ones.
-
4. Can I do it during my lunch break?
Absolutely. Exact treatment times may vary depending on the patient, but a single session typically takes no longer than 30 minutes.
-
5. How many sessions will I need to see results?
Most patients receive 6-8 treatments per area. The exact number will depend on each individual person and the area being treated.
-
6. How often/far apart should the treatments be?
Treatments are done once a week.
-
7. Do I need to do anything to prepare for the treatment?
Just make sure your skin is clean. Do not apply any lotions, creams or makeup before your treatment. Remove all jewelry around the area being treated.
-
1. How long does a treatment session last?
Each session lasts 15-30 minutes, depending on the area being treated.
-
2. What does the treatment feel like?
The applicator will slowly get warmer till your skin reaches the needed temperature and stays within that temperature range. But if you do feel it’s too hot, just let the operator know and they can easily lower the temperature a bit.
-
3. Will it be painful?
Treatments are comfortable. Some say it feels like a relaxing hot stone massage. Some even fall asleep during the treatment.
-
Treatments are comfortable. Some say it feels like a relaxing hot stone massage. Some even fall asleep during the treatment.
If anything, you’ll feel relaxed. The treatment is comfortable and has been compared to a relaxing hot stone massage. Some patients have even fallen asleep while they were getting treated.
-
2. How long will the results last and are they permanent?
The treatments are cumulative. After your final treatment, your results will continue to improve for up to 3-4 months. After that, your face and body continue to age, so maintenance is recommended once a month or every 3-4 months, depending on the person.
-
3. Is there really no downtime?
Absolutely no downtime. The treatment area may look a little flushed and warm to the touch, but this will go away within 1-2 hours. You can return to your daily schedule immediately after your treatment.
-
4. Do I need to do anything after my treatment?
No, you do not need to do anything after your treatment.
-
5. Can I put makeup up on right after my treatment?
Yes, you can apply makeup immediately after your treatment.
BEFORE THE TREATMENT
DURING THE TREATMENT
AFTER THE TREATMENT
Speak To An Expert
To book a consultation or a demo, please contact us by clicking the button below and a representative will reach out to you directly.
Why Venus
Body Devices
Body Treatments
Face & Skin Devices
Face & Skin Treatments
Hair Restoration Devices
Hair Removal Devices
Legal
For more information call: (888) 907-0115 // info@venusconcept.com // 235 Yorkland Blvd., Suite 900, Toronto, ON, M2J 4Y8 Canada
For more information call: (888) 907-0115 // info@venusconcept.com // 235 Yorkland Blvd., Suite 900, Toronto, ON M2J 4T8 Canada